Chinese Journal of Tissue Engineering Research ›› 2016, Vol. 20 ›› Issue (35): 5277-5283.doi: 10.3969/j.issn.2095-4344.2016.35.016
Previous Articles Next Articles
Expression of miR-140-3p in synovial fluid of knee osteoarthritis patients reflects the progression of osteoarthritis
Yin Chun-ming1, Pan Xiao-hua2
- 1Second Clinical Medical College of Jinan University, Shenzhen People’s Hospital, Shenzhen 518020, Guangdong Province, China; 2Department of Orthopedics and Traumatology, BaoAn Hospital Affiliated to Southern Medical University, Shenzhen 518100, Guangdong Province, China
-
Revised:2016-06-13Online:2016-08-26Published:2016-08-26 -
Contact:Pan Xiao-hua, M.D., Chief physician, Professor, Doctoral supervisor, Department of Orthopedics and Traumatology, BaoAn Hospital Affiliated to Southern Medical University, Shenzhen 518100, Guangdong Province, China -
About author:Yin Chun-ming, Studying for master’s degree, Second Clinical Medical College of Jinan University, Shenzhen People’s Hospital, Shenzhen 518020, Guangdong Province, China -
Supported by:The Project of Science and Technology Department of Shenzhen City in 2013, No. JCYJ20130402101926968; the International Cooperation Project of Shenzhen City in 2013, No. GJHZ20130418150248986
CLC Number:
Cite this article
Yin Chun-ming, Pan Xiao-hua. Expression of miR-140-3p in synovial fluid of knee osteoarthritis patients reflects the progression of osteoarthritis[J]. Chinese Journal of Tissue Engineering Research, 2016, 20(35): 5277-5283.
share this article
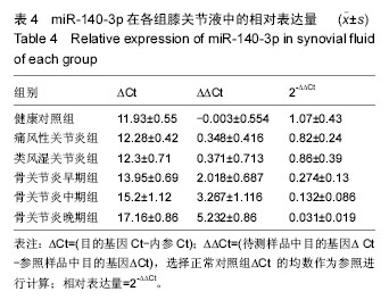
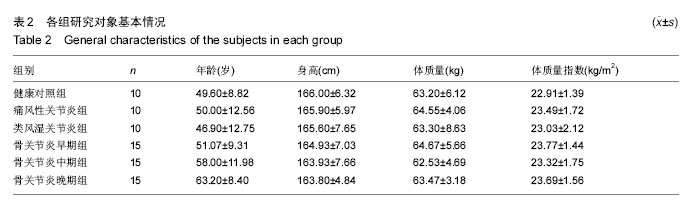
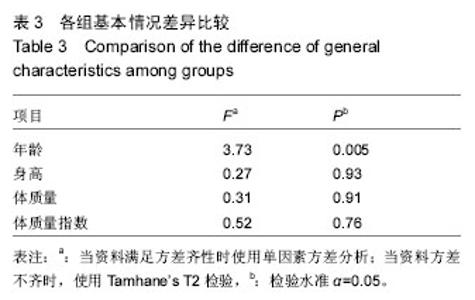
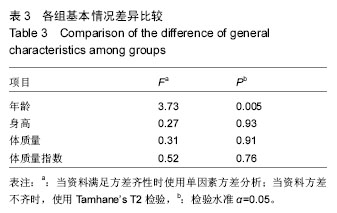
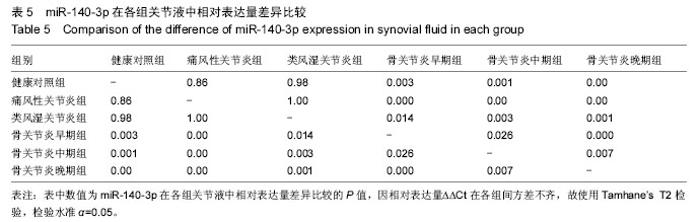
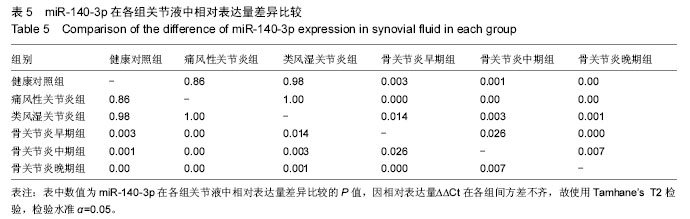
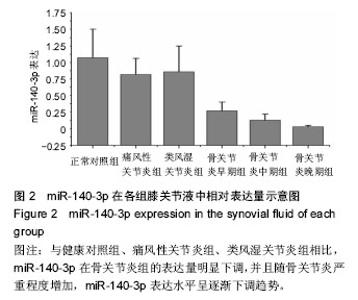
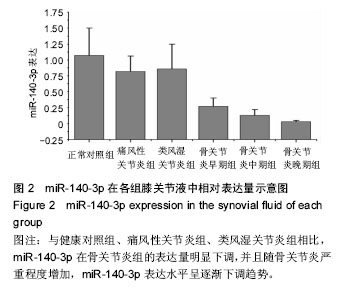
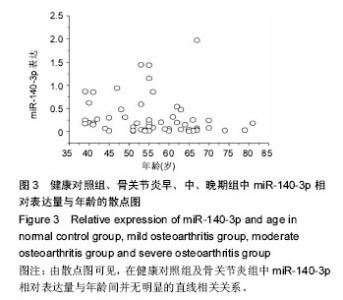
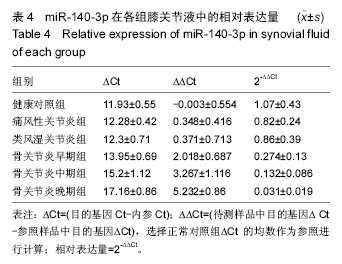
对健康对照组,类风湿关节炎组,痛风性关节炎组及骨关节炎早、中、晚期组患者膝关节液中miR-140-3p、miR-140-5p的相对表达量(表4)行Levene法方差齐性检验,结果显示各组方差不齐(Levene Statistic=13.4,P < 0.05)。 进一步对miR-140-3p在各组关节液中的相对表达量行Tamhane’s T2检验(表5),结果显示:miR-140-3p表达量在健康对照组、痛风性关节炎组与类风湿关节炎组间差异无显著性意义(P > 0.05);非骨关节炎组与骨关节炎组间相对表达量差异有显著性意义(P < 0.05),且miR-140-3p在非骨关节炎组中的表达量是骨关节炎组的9.6倍,健康对照组的表达量是骨关节炎组的11.4倍。miR-140-3p在骨关节炎早期、中期、晚期组间相对表达量差异均有显著性意义(表5),且随骨关节炎严重程度的增加miR-140-3p表达量呈相对减少的趋势(图2)。 将miR-140-3p在健康对照组,骨关节炎早、中、晚期组间的相对表达量进一步行Spearman等级相关分析,结果显示Spearman等级相关系数r=-0.87,P=0.00。按α=0.05水准,可以认为miR-140-3p的表达量与骨关节炎的严重程度间存在负相关关系。"
| [1] Lotz MK, Kraus VB. New developments in osteoarthritis. Posttraumatic osteoarthritis: pathogenesis and pharmacological treatment options. Arthritis Res Ther. 2010;12(3):211.
[2] Hashimoto M, Nakasa T, Hikata T, et al. Molecular network of cartilage homeostasis and osteoarthritis. Med Res Rev. 2008;28(3):464-481.
[3] Goldring MB, Marcu KB. Cartilage homeostasis in health and rheumatic diseases. Arthritis Res Ther. 2009;11(3):224.
[4] Goldring MB, Goldring SR. Articular cartilage and subchondral bone in the pathogenesis of osteoarthritis. Ann N Y Acad Sci. 2010;1192:230-237.
[5] Miyaki S, Sato T, Inoue A, et al. MicroRNA-140 plays dual roles in both cartilage development and homeostasis. Genes Dev. 2010;24(11):1173-1185.
[6] Tardif G, Hum D, Pelletier JP, et al. Regulation of the IGFBP-5 and MMP-13 genes by the microRNAs miR-140 and miR-27a in human osteoarthritic chondrocytes. BMC Musculoskelet Disord. 2009;10:148.
[7] Liang ZJ, Zhuang H, Wang GX, et al. MiRNA-140 is a negative feedback regulator of MMP-13 in IL-1β-stimulated human articular chondrocyte C28/I2 cells. Inflamm Res. 2012;61(5):503-509.
[8] Rodriguez A, Griffiths-Jones S, Ashurst JL, et al. Identification of mammalian microRNA host genes and transcription units. Genome Res. 2004;14(10A): 1902-1910.
[9] Nicolas FE, Pais H, Schwach F, et al. mRNA expression profiling reveals conserved and non-conserved miR-140 targets. RNA Biol. 2011;8(4):607-615.
[10] Hwang S, Park SK, Lee HY, et al. miR-140-5p suppresses BMP2-mediated osteogenesis in undifferentiated human mesenchymal stem cells. FEBS Lett. 2014;588(17):2957-2963.
[11] Huang W, Li MD. Nicotine modulates expression of miR-140*, which targets the 3'-untranslated region of dynamin 1 gene (Dnm1). Int J Neuropsychopharmacol. 2009;12(4):537-546.
[12] Takata A, Otsuka M, Kojima K, et al. MicroRNA-22 and microRNA-140 suppress NF-κB activity by regulating the expression of NF-κB coactivators. Biochem Biophys Res Commun. 2011;411(4):826-831.
[13] Tardif G, Pelletier JP, Fahmi H, et al. NFAT3 and TGF-β/SMAD3 regulate the expression of miR-140 in osteoarthritis. Arthritis Res Ther. 2013;15(6):R197.
[14] Karlsen TA, Jakobsen RB, Mikkelsen TS, et al. microRNA-140 targets RALA and regulates chondrogenic differentiation of human mesenchymal stem cells by translational enhancement of SOX9 and ACAN. Stem Cells Dev. 2014;23(3):290-304.
[15] Felson DT. The current and future status of biomarkers in osteoarthritis. J Rheumatol. 2014;41(5):834-836.
[16] Blanco FJ. Osteoarthritis year in review 2014: we need more biochemical biomarkers in qualification phase. Osteoarthritis Cartilage. 2014;22(12):2025-2032.
[17] Altman R, Asch E, Bloch D, et al. Development of criteria for the classification and reporting of osteoarthritis. Classification of osteoarthritis of the knee. Diagnostic and Therapeutic Criteria Committee of the American Rheumatism Association. Arthritis Rheum. 1986;29(8):1039-1049.
[18] Arnett FC, Edworthy SM, Bloch DA, et al. The American Rheumatism Association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum. 1988;31(3):315-324.
[19] Wallace SL, Robinson H, Masi AT, et al. Preliminary criteria for the classification of the acute arthritis of primary gout. Arthritis Rheum. 1977;20(3):895-900.
[20] Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods. 2001;25(4): 402-408.
[21] Pereira D, Severo M, Ramos E, et al. Potential role of age, sex, body mass index and pain to identify patients with knee osteoarthritis. Int J Rheum Dis. 2015. [Epub ahead of print]
[22] Felson DT, Lawrence RC, Dieppe PA, et al. Osteoarthritis: new insights. Part 1: the disease and its risk factors.Ann Intern Med. 2000;133(8):635-646.
[23] Felson DT, Nevitt MC. Epidemiologic studies for osteoarthritis: new versus conventional study design approaches. Rheum Dis Clin North Am. 2004;30(4): 783-797.
[24] Laxafoss E, Jacobsen S, Gosvig KK, et al. Case definitions of knee osteoarthritis in 4,151 unselected subjects: relevance for epidemiological studies: the Copenhagen Osteoarthritis Study. Skeletal Radiol. 2010;39(9):859-866.
[25] Chen LH, Chiou GY, Chen YW, et al. MicroRNA and aging: a novel modulator in regulating the aging network. Ageing Res Rev. 2010;9 Suppl 1:S59-66.
[26] Xu D, Takeshita F, Hino Y, et al. miR-22 represses cancer progression by inducing cellular senescence. J Cell Biol. 2011;193(2):409-424.
[27] Jazbutyte V, Fiedler J, Kneitz S, et al. MicroRNA-22 increases senescence and activates cardiac fibroblasts in the aging heart. Age (Dordr). 2013; 35(3):747-762.
[28] Liang R, Bates DJ, Wang E. Epigenetic Control of MicroRNA Expression and Aging. Curr Genomics. 2009;10(3):184-193.
[29] Lewis BP, Burge CB, Bartel DP. Conserved seed pairing, often flanked by adenosines, indicates that thousands of human genes are microRNA targets. Cell. 2005;120(1):15-20.
[30] Miyaki S, Asahara H. Macro view of microRNA function in osteoarthritis. Nat Rev Rheumatol. 2012;8(9):543-552.
[31] Ason B, Darnell DK, Wittbrodt B, et al. Differences in vertebrate microRNA expression.Proc Natl Acad Sci U S A. 2006;103(39):14385-14389.
[32] Wienholds E, Kloosterman WP, Miska E, et al. MicroRNA expression in zebrafish embryonic development. Science. 2005;309(5732):310-311.
[33] Tuddenham L, Wheeler G, Ntounia-Fousara S, et al. The cartilage specific microRNA-140 targets histone deacetylase 4 in mouse cells. FEBS Lett. 2006; 580(17):4214-4217.
[34] Miyaki S, Nakasa T, Otsuki S, et al. MicroRNA-140 is expressed in differentiated human articular chondrocytes and modulates interleukin-1 responses. Arthritis Rheum. 2009;60(9):2723-2730.
[35] Araldi E, Schipani E. MicroRNA-140 and the silencing of osteoarthritis. Genes Dev. 2010;24(11):1075-1080.
[36] Zhang R, Ma J, Yao J. Molecular mechanisms of the cartilage-specific microRNA-140 in osteoarthritis. Inflamm Res. 2013;62(10):871-877.
[37] Waldman SA, Terzic A. MicroRNA signatures as diagnostic and therapeutic targets. Clin Chem. 2008; 54(6):943-944.
[38] Murata K, Yoshitomi H, Tanida S, et al. Plasma and synovial fluid microRNAs as potential biomarkers of rheumatoid arthritis and osteoarthritis. Arthritis Res Ther. 2010;12(3):R86. |
| [1] | Shi Bin, An Jing, Chen Long-gang, Zhang Nan, Tian Ye . Influencing factors for pain after total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 993-997. |
| [2] | Wang Xian-xun. Impact of local compression cryotherapy combined with continuous passive motion on the early functional recovery after total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 998-1003. |
| [3] | Lu Yao-jia, Xiong Chuan-zhi, Li Xiao-lei, Hu Han-sheng, Chen Gang, Wang Qiang, Lu Zhi-hua. Comparison of two methods for reducing blood loss during total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 1004-1008. |
| [4] | Yuan Wei, Zhao Hui, Ding Zhe-ru, Wu Yu-li, Wu Hai-shan, Qian Qi-rong. Association between psychological resilience and acute mental disorders after total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 1015-1019. |
| [5] | Chen Qun-qun, Qiao Rong-qin, Duan Rui-qi, Hu Nian-hong, Li Zhao, Shao Min. Acu-Loc®2 volar distal radius bone plate system for repairing type C fracture of distal radius [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 1025-1030. |
| [6] | Huang Xiang-wang, Liu Hong-zhe. A new low elastic modulus of beta titanium alloy Ti2448 spinal pedicle screw fixation affects thoracic stability: biomechanical analysis [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 1031-1035. |
| [7] | Xie Qiang. Three-dimensional finite element model for biomechanical analysis of stress in knee inversion and external rotation after posterior cruciate ligament rupture [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 1036-1040. |
| [8] | He Ze-dong, Zhao Jing, Chen Liang-yu, Li Ke, Weng Jie. Multilevel finite element analysis on the biological tribology damage of water on bone tissue [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 1041-1045. |
| [9] | Jiang Zi-wei, Huang Feng, Cheng Si-yuan, Zheng Xiao-hui, Sun Shi-dong, Zhao Jing-tao, Cong Hai-chen,Sun Han-qiao, Dong Hang. Design and finite element analysis of digital splint [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 1052-1056. |
| [10] | Wang Fei, Liu Zhi-bin, Tao Hui-ren, Zhang Jian-hua, Li Chang-hong, Cao Qiang, Zheng Jun, Liu Yan-xiong, Qu Xiao-peng. Clinical efficacy of preoperative osteotomy designs using paper-cut technology versus photoshop software for ankylosing spondylitis with kyphosis [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 1057-1063. |
| [11] | Li Hui, Ma Jun-yi, Ma Yuan, Zhu Xu . Establishment of a three-dimensional finite element model of ankylosing spondylitis kyphosis [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 1069-1073. |
| [12] | Ling Guan-han, Ou Zhi-xue, Yao Lan, Wen Li-chun, Wang Guo-xiang, Lin Heng-feng. Establishment of simulating three-dimensional model of China-Japan Friendship Hospital Classification for L type osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 1074-1079. |
| [13] | Fu Wei-min, Wang Ben-jie. Assessing the degree of necrotic femoral head, and association of blood supply with pathlogical changes: study protocol for a diagnostic animal trial [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 1086-1091. |
| [14] | Zhang Wen-qiang, Ding Qian, Zhang Na. Associations between alpha angle and herniation pit on oblique axial magnetic resonance imaging in asymptomatic hip joints of adults [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 1098-1103. |
| [15] | Sun Xiao-xin1, Zhou Wei2, Zuo Shu-ping3, Liu Hao1, Song Jing-feng1, Liang Chun-yu1. Morphological characteristics for the magnetic resonance imaging assessment of discoid lateral meniscal tears in children [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 1104-1109. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||